B-naphthylamine isopac
B-naphthylamine isopac
CAS: 91-59-8
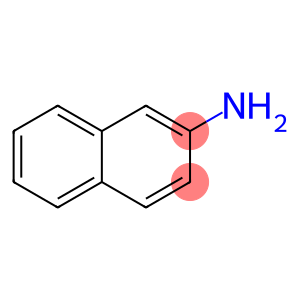
Molecular Formula: C10H9N
B-naphthylamine isopac - Names and Identifiers
| Name | B-naphthylamine isopac |
| Synonyms | l-Naphthylamine 6-Naphthylamine 2-Aminonaphthalene naphthalen-2-amine B-naphthylamine isopac 2-naphthylaminemustard 2-NAPHTHYLAMINE OEKANAL, 100 MG 2-Aminonaphthalene, b-Naphthylamine |
| CAS | 91-59-8 |
| EINECS | 202-080-4 |
| InChI | InChI=1/C10H9N/c11-10-6-5-8-3-1-2-4-9(8)7-10/h1-7H,11H2 |
| InChIKey | JBIJLHTVPXGSAM-UHFFFAOYSA-N |
B-naphthylamine isopac - Physico-chemical Properties
| Molecular Formula | C10H9N |
| Molar Mass | 143.19 |
| Density | 1.061g/mLat 25°C(lit.) |
| Melting Point | 111-113°C(lit.) |
| Boling Point | 306°C(lit.) |
| Solubility | Solubility Soluble in hot water, ethanol, ether |
| Vapor Presure | 2.56 x 10-4 mmHg at 20–30 °C (quoted, Mercer et al., 1990) |
| Appearance | powder |
| Color | pink to purple |
| Exposure Limit | Since it is a carcinogen, there is no TLV TWA for this compound. Recognized Carcinogen (ACGIH); Carcinogen (OSHA);Human Sufficient Evidence (IARC). |
| Merck | 13,6425 |
| BRN | 3939429 |
| pKa | 4.16(at 25℃) |
| Storage Condition | -20°C Freezer |
| Stability | Stable. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.5000 (estimate) |
| Physical and Chemical Properties | density 1.061
|
B-naphthylamine isopac - Risk and Safety
| Risk Codes | R45 - May cause cancer R22 - Harmful if swallowed R51/53 - Toxic to aquatic organisms, may cause long-term adverse effects in the aquatic environment. |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S61 - Avoid release to the environment. Refer to special instructions / safety data sheets. |
| UN IDs | UN 1650 6.1/PG 2 |
| WGK Germany | 3 |
| RTECS | QM2100000 |
| HS Code | 29214500 |
| Hazard Class | 6.1(a) |
| Packing Group | II |
| Toxicity | LD50 (intraperitoneal) for mice 200 mg/kg (quoted, RTECS, 1985). |
B-naphthylamine isopac - Reference Information
| pH indicator color change ph range | Non & uorescence (2.8) to violet & uorescence (4.4) |
| Henry's Law Constant | 2.01 x 10-9 atm?m3/mol at 25 °C (quoted, Mercer et al., 1990) |
| main applications | Microelectronics, power transmission & fluid, insulators for electronic devices, photoresists, display device, imaging process, semiconductors, diesel fuel additives, battery, adhesive, paints, inks, chalk, leather, textiles, dye synthesis, soil products |
| NIST chemical information | Information provided by: webbook.nist.gov (external link) |
| (IARC) carcinogen classification | 1 (vol. 4, sup 7, 99, 100f) 2012 |
| EPA chemical information | Information provided by: ofmpub.epa.gov (external link) |
| The health hazards of naphthylamine | Naphthylamine has two isomers, namely methyl naphthylamine (1-naphthylamine or α naphthylamine) and ethyl naphthylamine (2-naphthylamine or β naphthylamine), molecular weight 143.18. Methylnaphthylamine is a yellow needle-like crystal with a specific gravity of 1.13, a melting point of 50 ℃, and a boiling point of 300.8 ℃; it is easily soluble in ether and alcohol, and slightly soluble in water. Ethylnaphthylamine is colorless or pink flake crystal with 1.061 specific gravity, melting point 110.2 ℃, boiling point 306.1 ℃; Soluble in hot water, alcohol, ether and benzene. Naphthylamine is mainly used in the dye industry and is used as an accelerator for rubber vulcanization. Methylnaphthylamine is also used to synthesize some compounds containing naphthyl groups. Both are intermediates in organic synthesis such as dyes. The production of 1-naphthylamine often coexists with about 5% 2-naphthylamine. Large-scale occupational disease surveys in the 2050s believed that 2-naphthylamine is a strong carcinogen, and 1-naphthylamine is also carcinogenic, mainly because its metabolites cause bladder cancer and can also cause other cancers. It is contained in both coal tar and cigarette smoke. At present, most countries have banned the production of 2-naphthylamine and restricted the content of 2-naphthylamine coexisting in 1-naphthylamine. naphthylamine can enter the human body through the respiratory tract, and can also enter from the gastrointestinal tract and skin. After naphthylamine enters the human body, a small part is excreted from urine in its original form, and most of it is transformed into hydroxy derivatives and quinone imine (NH:C6H4:O) derivatives with carcinogenic effects. Ethylnaphthylamine can cause contact dermatitis. Acute poisoning can lead to methemoglobinemia or acute hemorrhagic cystitis. The main hazard of long-term exposure to ethylnaphthylamine is the occurrence of bladder tumors, with an average induction period of 15 to 20 years. A small number of contacts can only get sick several years after being transferred from work. Most of them are malignant bladder cancer with slow onset. The early symptoms are sudden painless hematuria or microscopic hematuria. Cystoscopy can assist in the diagnosis. For long-term contacts, routine urine and cytological examination should be carried out regularly. If there is any abnormality, cystoscopy should be carried out in time. After confirming the diagnosis of bladder tumor, surgical treatment should be performed immediately. Some countries believe that naphthylamine is a human carcinogen, so it is stipulated that naphthylamine is not allowed to be detected in the workplace. Our country requires strict control, but does not make specific provisions. Reference: Chinese Medical Encyclopedia. |
| Use | The amino group of naphthylamine reacts with nitrous acid to form diazonium salt, and can be converted into a variety of naphthalene derivatives, which are used to make dyes and organic synthesis. It is also used as an organic analysis reagent and fluorescence indicator. |
| Production method | Since naphthalene nitration cannot obtain 2-nitronaphthalene, the production method is different from 1-naphthalene. 2-Naphthylamine is derived from 2-naphthol by pressurized ammonolysis. |
| category | toxic substances |
| toxicity classification | poisoning |
| acute toxicity | oral administration-rat LD50: 727 mg/kg; Abdominal cavity-mouse LD50: 200 mg/kg |
| flammability hazard characteristics | open flame is combustible; heated to release toxic nitrogen oxide gas |
| storage and transportation characteristics | warehouse ventilation and low temperature drying; Store separately from acids, oxidants and food additives |
| fire extinguishing agent | foam, carbon dioxide, dry powder, sand |
| toxic substance data | information provided by: pubchem.ncbi.nlm.nih.gov (external link) |
Last Update:2024-04-09 21:01:54
Supplier List
Spot supply
Product Name: Dapoxetine Impurity 113 Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 2-Naphthylamine Request for quotation
CAS: 91-59-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 91-59-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-naphthylamine Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: β-Naphthylamine Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Dapoxetine Impurity 113 Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: 2-Naphthylamine Request for quotation
CAS: 91-59-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 91-59-8
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Spot supply
Product Name: 2-naphthylamine Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Spot supply
Product Name: β-Naphthylamine Visit Supplier Webpage Request for quotationCAS: 91-59-8
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
View History



